When a chemical reaction takes place, atoms in a starting substance rearrange themselves and join back together to form a new substance. But, the number of atoms will still be the same before and after the reaction.
As in in the chemical reaction below:
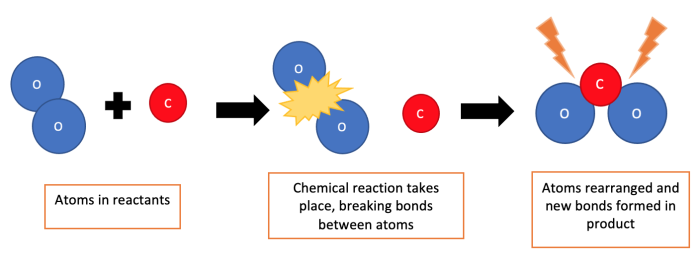
This is what happens in all chemical reactions. Let's now think about the reaction we see when a piece of potassium reacts with water:
.png)
It's a pretty spectacular result - the potassium bursts into flames - purple flames in fact!
The word equation is:
Potassium + Water → Potassium hydroxide + Hydrogen
As with the previous chemical reaction, the atoms in the reactants will just break apart and reform into a different order to form the products. So this means that if we know the mass of the potassium before the experiment, the mass of water that it reacted with, and the mass of the potassium hydroxide in the product, we could work out how much hydrogen gas has also been produced. It's just basically a little maths equation.
Potassium + Water → Potassium hydroxide + Hydrogen
5 g + 20 g → 15 g + ?
The mass of hydrogen produced would be 25 g - 15 g = 10 g
As you can see, the mass of the two substances before is the same as the substances produced! This is called the Law of Conservation of Mass.
Let's look at some examples in the questions that follow.








